Tuberous Sclerosis Complex (TSC) identified by genetic testing after histologicdiagnosis of Chromophobe Renal Cell Carcinoma
junio 15th, 2023

** Division of Urology, San Jose Hospital Oncology Center, Hermosillo Mexico
¶ Associate Professor of Urology, St. Joseph’s Hospital, London Ontario, Canada
*Department of Pathology, Advanced Pathology, Hermosillo Mexico
≠Division of Oncology, San Jose Hospital Oncology Center, Hermosillo Mexico
†Assistant Professor, Department of Urology, University of Arizona College of Medicine, Tucson Arizona
§ Professor and Chief of Urology, University of Arizona College of Medicine, Tucson Arizona
We report the case of a female patient with a histologic diagnosis of Chromophobe renal cell carcinoma after a left laparoscopic radical nephrectomy. Due to the unusual histologic diagnosis a genetic test was ordered identifying tuberous sclerosis disease.
Tuberous sclerosis complex (TSC) was described in the 19th Century by von Recklinghausen (1), it’s an autosomal dominant (2), multi-organic disease that can affect skin, brain, kidneys lungs and heart replacing normal parenchyma by different cell types. Development of tumors in TSC follow the inactivation of TSC1 (encoding Hamartin), or TSC2 (encoding Tuberin)(4, 9) genes(2) which lead to alterations of the TSC1-TSC2 intracellular protein complex, causing overactivation of the mammalian target of rapamycin (mTOR) protein complex (3), which is the cause for the development of tumors in different organs(4). TSC has an incidence of 1/6000 to 1/10,000 live births.
Chromophobe renal cell carcinoma (ChRCC) is the third most common renal cancer after clear cell and papillary renal cell carcinoma comprising 5% of all renal tumors, with an incidence of metastatic disease of 7% (5); Casuscelli et al identified enrichment of TP53 and PTEN mutations in metastatic ChRCC(7). Under-expression of gamma-glutamil transferase 1 (GGT1) has been identified as a risk factor for the development for ChRCC(5). Oncocytomas and ChRCC share a common origin in the collecting system which can difficult a histological diagnosis(6); looking for deletions in RB1 and ERBB4 which subsequently distinguishes ChRCC from oncocytomas(6) . ChRCC can occur in two autosomal dominant genetic alterations: Birt-Hogg-Dube syndrome and tuberous sclerosis complex(5).
Tuberous sclerosis complex-associated renal cell carcinoma (TSC-RCC) has been identified as a subtype of RCC(8, 2). It is associated with TSC, female predominance, young age at onset and indolent evolution(8). TSC-RCC can lead to the development of angiomyoadenomatous tumors(2,8), TSC-associated papillary RCC, ChRCC or oncocytic/chromophobe tumors, eosinophilic/macrocystic and unclassified RCC(8) . The mTOR pathway activation and TSC2 mutations have been identified in TSC-RCC(8).
A 39 year old female patient with a history of gestational diabetes and hypothyroidism for four years, with an incidental finding on abdominal CT scan ordered for gastric complaints, demonstrated presence of two contrast enhancing tumors on the left kidney, one in the posterior interpolar region measuring 2 x 2 cms, and another one in the lower pole measuring 3.5 x 3.2 cms (Fig. 1). Due to presence of multifocal tumors a left laparoscopic radical nephrectomy was performed, with a histologic diagnosis of a multicentric Fuhrman 3, chromophobe renal cell carcinoma, with other nodules composed of oncocytic cells disposed in groups, and a capsular leiomyoma (Fig. 2). Due to the unusual histologic diagnosis a genetic profile was ordered, together with follow-up labs, chest x-ray and abdominal ultrasound. Genetic sequence analysis and deletion/duplication testing of 83 genes (Invitae Multi-Cancer Panel) was performed by Invitae Genetic Testing (1400 16th Street, San Francisco, CA 94103) reporting a pathogenic heterozygous variant, c.2605A>T (p.Lys869*) identified in the TSC1 gene, consistent with a diagnosis of tuberous sclerosis complex (TSC). With this result a genetic testing was ordered on her offspring and was scheduled for follow-up visits.
Tuberous sclerosis complex is highly variable in clinical presentation. Elements required for a proper diagnosis includes genetic and clinical criteria. It is recommended to identify mutations in TSC1 and TSC2 regardless of clinical findings(1). Clinical diagnostic criteria include dermatologic and dental, ophthalmic, neurological(4) , cardiovascular, pulmonary, endocrine, gastrointestinal and renal alterations(1, 3). Kidney angiomyolipomas are frequently associated with TSC and have been observed in up to 80% of these patients, and they can be observed in other organs as well (1, 2). Although multiple renal cysts are infrequent in the general population they are relatively common in TSC as the TSC2 and PKD1 genes are adjacent and transcribed in opposite directions on the same chromosome, and both genes can present deletions. RCC and oncocytoma(9) in TSC patients has been recognized for many years and they develop by inactivation of TSC2 (2). The renal tumors are often multifocal(9) and it was not clear if patients with multiple RCCs had intrarenal metastasis of a single tumor clone or multifocal tumor development(2), however the recent understanding of the genetic pathogenesis of TSC related RCC conclude that RCCs arise independently(2) .
Chromophobe renal cell carcinoma is an uncommon variant and if identified without a previous history of genetic problems, the patient should be referred for genetic counselling in order to rule out involvement of other organs.
Fig. 1:
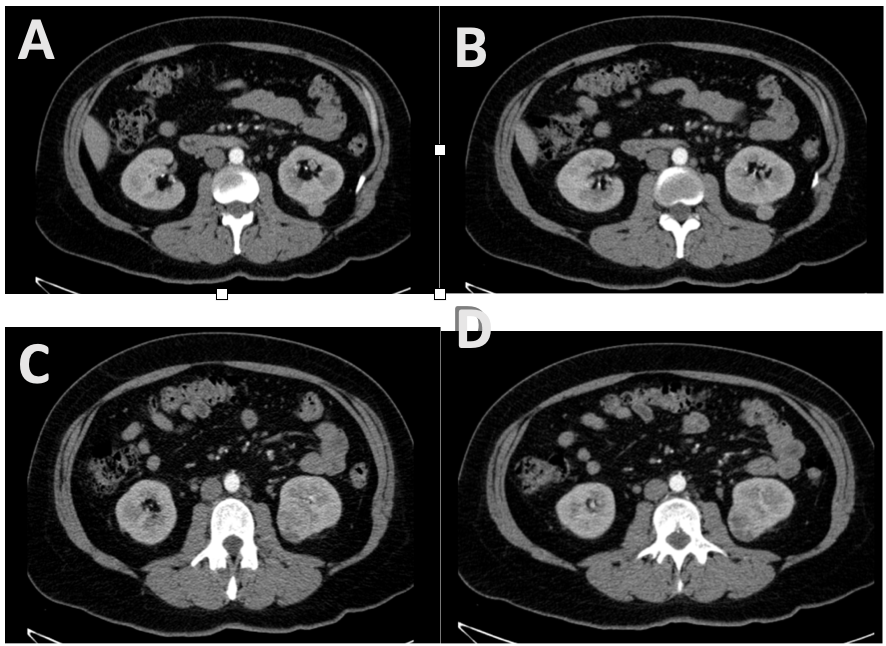
Figure 1.- A and B: An enhancing, smooth walled, round tumor in the left kidney’s posterior capsular interpolar region. C and D: Left lower pole heterogeneous, strongly enhancing mass arising from the cortex.
Fig. 2:
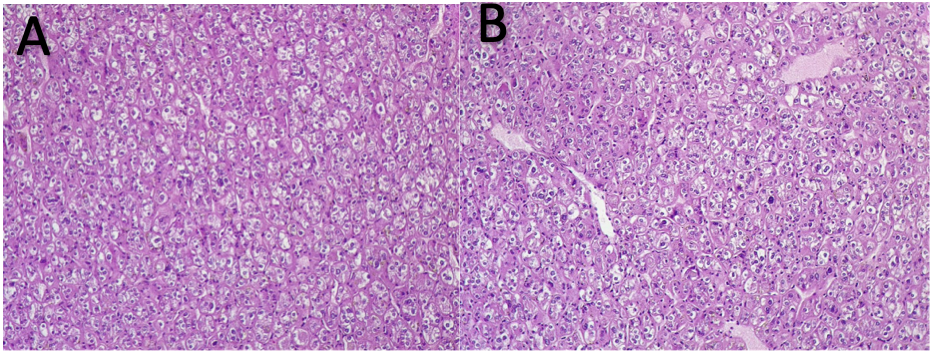
Figure 2.- A: Classic type kidney chromophobe carcinoma with polygonal cells with clear and finely reticular cytoplasm with perinuclear halo and with pleomorphic cells. B: Classic type kidney chromophobe carcinoma with cells with central or eccentric nucleus with dense chromatin with cellular disposition around blood vessels.
1.- Northrup H, Krueger DA. Tuberous sclerosis complex diagnostic criteria update: Recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediatric Neurology 49 (2013) 243-254.
2.- Lam HC, Nijmeh J, Henske EP. New developments in the genetics and pathogenesis of tumours in tuberous sclerosis complex. J Pathol 2017; 241: 219-225.
3.- Curatolo P, Moavero R, e Vries PJ. Neurological and neuropsychiatric aspects of tuberous sclerosis complex. The Lancet Neurology. Vol 14, Issue 7, July 2015, 733-745.
4.- Slowinska M, Jozwiak S, Peron A, Borkowska J, Chmielewski D, Sadowski K, Jurkiewicz E, Vignoli A, La Briola F, Canevini MP, Kotulska-Jozwiak K. Early diagnosis of tuberous sclerosis complex: a race agains time: How to make the diagnosis before seizurse? Orphanet Journal of Rare Diseases (2018) 13:25.
5.- Priolo C, Khabibullin D, REznik E, Filippakis H, Ogorek B, Kavanagh TR, Nijmeh J, Herbert ZT, Asara JM, Kwiatkowski DJ, Wu CL, Heske EP. Impairment of gamma-glutamyl transferase 1 activity in the metabolic pathogenesis of chromophobe renal cell carcinoma. PNAS June 2018, vol 115 no. 27.
6.- Liu Q, Cornejo KM, Cheng L, Hutchinson L, Wang M, Zhang S, Tomaszewics K, Cosar EF, Woda BA, Jiang Z. Next-generation sequencing to detect deletion of RB1 and ERBB4 genes in chromophobe renal cell carcinoma. A potential role in distinguishing chromophobe renal cel carcinoma from renal oncocytoma. Am J Pathol vol 188, No. 4, April 2018.
7.- Casuscelli J, Weinhold N, Gundem G, Wang L, Zabor EC, Drill E, Wang PI, Nanjangud GJ, Redzematovic A, Nargund AM, Manley BJ, Arcila ME, Donin NM, Cheville JC, Houston Thompson R, Pantuck AJ, Russo P, Cheng EH, Lee W, Tickoo SK, Ostrovnaya I, Creighton CJ, Papaemmanuil E, Seshan VE, Hakami AA, Hsieh JJ. Genomic landscape and evolution of metastatic chromophobe renal cell carcinoma. JCI Insight. 2017; 2(12): e92688.
8.- Park JH, Lee C, Chang MS, Kim K, Choi S, Lee H, Lee HS, Moon KC. Molecular characterization and putative pathogenic pathways of tuberous sclerosis complex-associated renal cell carcinoma. Translational Oncology. Vol. 11, No. 4. August 2018. 962-970.
9.-Bonsib SM, Boils C, Gokden N, Grignon D, Gu X, Higgins JPT, Leroy X, McKenney JK, Nasr SH, Phillips C, Sangoi AR, Wilson J, Zhang PL. Tuberous sclerosis complex: Hamartin and tuberin expression in renal cysts and its discordant expression in renal neoplasms. Pathol Res Pract 212 (2016) 972-979.
Alfonso Fernandez: urologiafernandez@gmail.com
Stephen E. Pautler: Stephen.Pautler@sjhc.london.on.ca
Minor Cordero: minorcb@gmail.com
Enrique Avila Monteverde: avilamonteverde@msn.com
Juan Chipollini: jchipollini@surgery.arizona.edu
Benjamin R. Lee: brlee@surgery.arizona.edu